Explain the Difference Between Filtration and Decantation
View the full answer. Difference Between Decantation and Filtration.

Difference Between Decantation And Filtration Compare The Difference Between Similar Terms
The major difference between decantation and filtration is in the process by which it is achieved.

. Experts are tested by Chegg as specialists in their subject area. In decantation the separation of two substances either of a solid and liquid or two immiscible liquids occurs by allowing the mixture to settle and separate. There are some mixtures which contain insoluble solid particles suspended in a liquid.
In filtration you are just dumpinf a mixed amount of substances through some pours and in ten seconds you are done. Decantation is pouring out of upper clear layer of liquid into another container to separate two immiscible liquids. 100 2 ratings Filtration is the direct separation of a solid precipitate and the solvent in which it is contained by th.
Decantation and filtration differ in the manner through which the methods separate substances in a mixture. In decantation when we pour out supernatant liquid there are chances of their getting mixed up with solute particles. FILTRATION IS faster.
The key difference between sedimentation and decantation is that the sedimentation allows the separation of two substances via settling of one substance whereas the decantation allows the separation of two substances via pouring off one substance. Sedimentation Decantation and Filtration. While both filtration and decantation can be used to separate impurities from liquids there are differences between them.
The key difference between decantation and filtration is that decantation separates two components in a mixture by pouring off one component whereas filtration separates two components by filtering off one component. The pouring out of a liquid from a vessel without disturbing the sediments is called decantation. Filtration on the other hand is a process of separating solids from liquids by passing it through a medium only a liquid can pass.
Once the substances have separated the lighter substance is poured out leaving the heavier. Explain the difference between filtration and decantation see the General laboratory Equipment and Procedures section of this manual. The major difference between decantation and filtration is in the process by which it is achieved.
By means of decantation you have to slowly. Why might one want to use filtration in this experiment rather than decantation. It cannot be used to separate two liquids.
This is done by pouring out the clear upper layer of liquid. Filtration is a better technique than sedimentation and decantation because it can separate very Fine insoluble particles as compared to the latter. Both sedimentation and decantation are important separation methods in analytical chemistry.
Decantation is a process for the separation of mixtures of immiscible liquids and solid such as suspension. Decantation is pouring away a liquid from solid impurities which have. Decantation is a process for the separation of mixtures of immiscible liquids and solid such as suspension.
A mixture of sand and water contains sand particles suspended in water. It is defined as the separation process in which two immiscible liquids are separated. Like mud settles from muddy water.
Why might one want to use filtration in this experiment rather than decantation. Filteration is a better process than decantation. Both decantation and filtration separate two components in a liquid-solid mixture or a mixture of two immiscible liquids under.
We review their content and use your feedback to keep the quality high. Difference Between Filtration and Decantation Explained Filtration. In decantation the mixture is not disturbed for few minutes and then the liquid is poured into.
Separation of a mixture of oil from water is an example of decantation. See full answer below. The key difference between decantation and filtration is that decantation separates two components in a mixture by pouring off one component whereas filtration separates two components by filtering off one component.
Filtration is the direct separation of the entire solution through a filter where the solid is trapped by the filter allowing the liquid to pass through. The solid particles which are insoluble in a liquid can be separated by decantation. Filtration is the process of separating insoluble impurities from a solution.
Filtration can be described as an act or process of filtering or cleaning something. This process can also be used to separate two liquids that do not mix eg oil and water. Why might one want to use filtration in this experiment rather than decantation.
Besides the filtrate obtained is generally pure solvent. Decantation is the method of cleaning in which we dont use a medium which has. Filtration on the other hand is a process of separating solids from liquids by passing it through a medium only a liquid can pass.
Correct answer to the question Explain the difference between filtration and decantation see the general laboratory equipment and procedures section of this manual. Like draining oil from a mixture of oil and water after allowing the two to separate and form distinct layers. Explain the difference between filtration and decantation see the General laboratory Equipment and Procedures section of this manual.

Difference Between Filtration And Decantation Explained

Difference Between Decantation And Filtration Compare The Difference Between Similar Terms
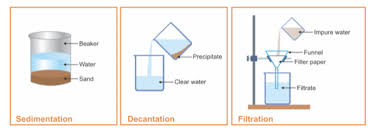
What Is Sedimentation Decantation And Filtration Home Work Help Learn Cbse Forum

No comments for "Explain the Difference Between Filtration and Decantation"
Post a Comment